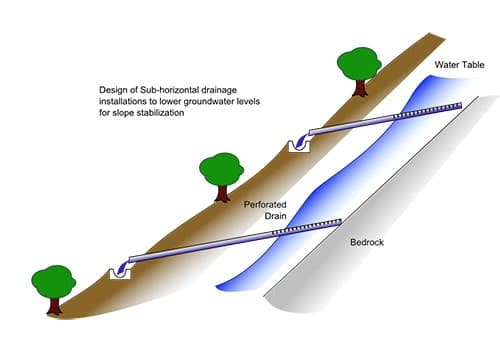
In geology, evaporites are water-soluble, mineral sediments that result from the evaporation of restricted bodies of water on the Earth’s surface. They are sedimentary rocks. Some evaporite deposits are highly valued. For example, halites (NaCl) can form diapirs (salt domes) where petroleum deposits can be trapped, nitrates are used in the manufacture of fertilizers and explosives, and potash (sylvite, KCl) is an important fertilizer.
Minerals in evaporite rocks include carbonates such as calcite (CaCO3), dolomite (CaMg(CO3)2, magnesite (MgCO3), and aragonite (CaCO3) which has a different crystal structure to calcite. Also found in evaporites are sulphates – anhydrite (CaSO4), gypsum (CaSO4.2H2O) and polyhalite (K2Ca2Mg(SO4)6.H2O). The chlorides include halite (NaCl), sylvite (KCl), and carnallite (KMgCl3.6H2O), as well as various borates, silicates, nitrates, and sulphocarbonates. Evaporite deposits occur in both marine and nonmarine sedimentary successions. Excluding the carbonates, less than 5% of all evaporites are classified as sulphate type, and greater than 95% are chloride type. Rock salt is the most common evaporite composed entirely of halite (NaCl), with a mineral content of 90–98% by volume, and subordinate amounts of anhydrite (CaSO4) and/or polyhalite (K2Ca2Mg(SO4)6.H2O).
All bodies of water on the Earth’s surface and in aquifers contain dissolved salts. To form minerals from these salts, the water must evaporate into the atmosphere, so that the minerals precipitate out. For this to happen, a water body must be in an environment where water input is less than the rate of evaporation. When evaporation occurs, the remaining water is enriched in salts, which precipitate when their concentration in water becomes high enough that they can no longer remain in solution. The less soluble a mineral is, the more readily it precipitates out of solution. The order of precipitation for several minerals is:
Most evaporite formations do not contain more than a few percent of evaporite minerals, the remainder being composed of the more typical detrital clastic rocks and carbonates.
This sequential precipitation is responsible for the colourful rhythmic patterns often associated with evaporite deposits.